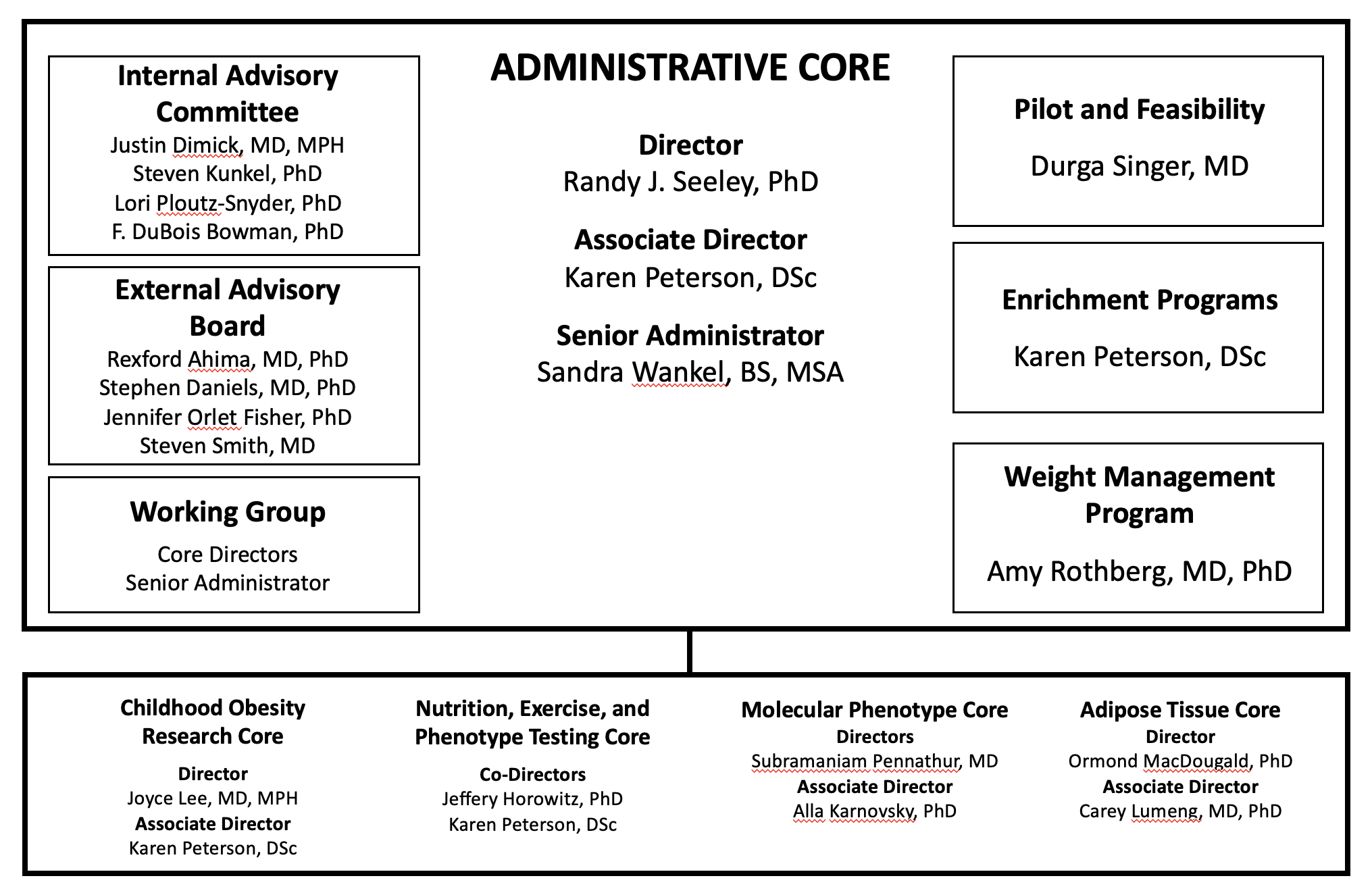
Administrative Core
The Administrative Core supports research and services in the five research cores and is responsible for the overall management of the MNORC.
Nutrition, Exercise and Phenotype Testing (NExT) Core
The NExT is a collaborative network of laboratory and research facilities on campus, which provide expanded support for clinical and translational research studies utilizing nutrition interventions and/or focused on obesity or obesity-related metabolic disorders in humans.
Childhood Obesity Research Core
The Childhood Obesity Research Core (CORC) supports basic, clinical, and translational researchers focused on childhood nutrition and obesity and enhances integration of research into the clinical delivery system.
Molecular Phenotyping Core (MPC)
The MPC performs functional metabolic studies in in vitro systems and provides analytical tools which permit structural identification and quantification of metabolites.
Adipose Tissue Core
The University of Michigan Adipose Tissue Core brings together laboratories across campus to research diabetes, obesity, and adipocyte biology.
Weight Management Program
The Weight Management Program is a Demonstration Unit that provides state of the art clinical care for patients with obesity. Patients can opt into a translational research program in which they undergo extensive baseline and interval biological and behavioral phenotyping during weight loss in order to determine their metabolic and neuro-psychosocial phenotype. Over 1,300 individuals have participated and 48,000 biological samples are available to investigators interested in nutrition, obesity and metabolism.
If you want to know what samples or data are available or have questions regarding procurement for your studies, go to research.med.umich.edu/biorepository