The Michigan Diabetes Research Center (MDRC) awarded Rose C. and Nathan L. Milstein Family Emerging Scholar Stephanie Eid, Ph.D., a Pilot/Feasibility Grant to investigate the roles of Schwann cells and extracellular vesicles in diabetic neuropathy.

"Pilot grants are incredibly important, especially to young investigators like me, because when you have promising initial data, a pilot grant can help you accelerate the pace of your research,” explained Eid. “In fact, that’s the point of them—to collect enough preliminary data to serve as a basis for a larger grant.”
What will Dr. Eid do with this support?
First, it is important to understand the problem she is fighting. Diabetes has become a major public health burden affecting over 537 million cases worldwide, with type 2 diabetes accounting for 90-95% of those cases. Type 2 diabetes is increasing in parallel with the rise in obesity with a projected 783 million global cases by 2045. Additionally, the incidence of prediabetes, a condition of impaired glucose that doesn’t yet meet the threshold for diabetes, is also increasing with over 541 million cases worldwide.
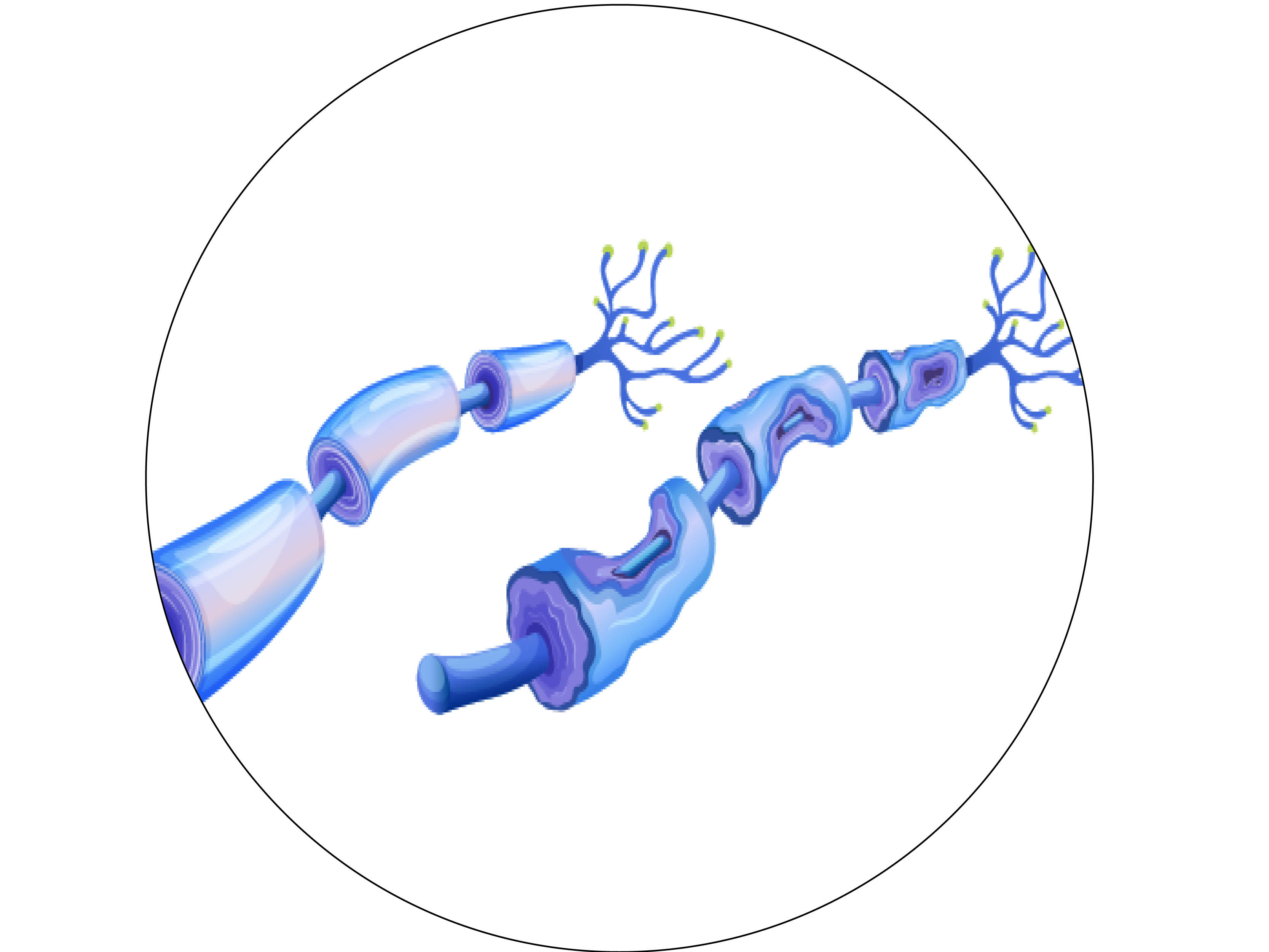
Both prediabetes and type 2 diabetes are associated with a host of debilitating long-term complications, including peripheral neuropathy, which develops in 30% of prediabetes patients and 60% of type 2 diabetes patients. Peripheral neuropathy is nerve damage beginning in the hands and feet that progresses up the limbs, leading to chronic pain, and can also cause non-healing ulcers and amputations. More than $10 billion in healthcare costs are attributed to peripheral neuropathy. There is currently no effective treatment, and glucose control has very little effect on peripheral neuropathy in these groups.
It is not all doom and gloom. Studies in the lab of the NeuroNetwork for Emerging Therapies and other groups have implicated metabolic syndrome (obesity, dyslipidemia, etc.) as a peripheral neuropathy risk factor, independent of glycemic status. This has led to the American Diabetes Association recommending lifestyle interventions to improve overall metabolic health, including a healthier diet. This has been shown to lessen peripheral neuropathy, but many, if not most, prediabetes and type 2 diabetes patients are unwilling or unable to engage in these dietary interventions. How do we help them?
That is where Dr. Eid’s work comes in. She believes that by understanding the mechanisms by which dietary interventions improve nerve function, which have yet to be studied, new therapy targets could be uncovered to help those who are unwilling or unable to engage in lifestyle changes.
How will she do it? First, Dr. Eid will look at the cellular interactions that are happening in the nerve microenvironment, or the nerve itself, particularly axons, and all the cells they are interacting with, in pre-clinical models. Particularly she will be studying Schwann cells, which support nerves by not only surrounding them to offer protection but also by ensuring that they receive the energy they need to function properly. When this interaction is compromised during diabetes or any kind of nerve injury, it leads to energetic failure.
Schwann cells, like other cells in the body, release extracellular vesicles, a type of cell that is taken up by axons. Dr. Eid wants to study the content of these extracellular vesicles in healthy and disease conditions to determine how they are either aiding nerves to meet their energy demands or promoting injury and neurodegeneration.
“We want to see if these vesicles contain injurious material that is being transferred to the axons and causing damage in prediabetes. We are also looking at the effect of diet because we know that a good dietary intervention might reverse neuropathy. We want to see whether this happens through the extracellular vesicles delivering healthy cargo to the axons, which restores insulin signaling to these nerves. This would offer us a potential new therapeutic target for peripheral neuropathy.
The second aim of the grant Dr. Eid describes as more “mechanistic.” After mimicking prediabetic conditions in Schwann cells in vitro, she examined the content of the extracellular vesicles and identified an increase in the expression of a particular microRNA, a small RNA molecule regulating gene expression.
Dr. Eid explains: “We think that this overly expressed microRNA is regulating and inducing insulin resistance. We want to see whether this is true, and then try to manipulate its expression see whether we can restore nerve function and improve insulin sensitivity. If that is possible, the possibilities of therapy targets are truly exciting.”
