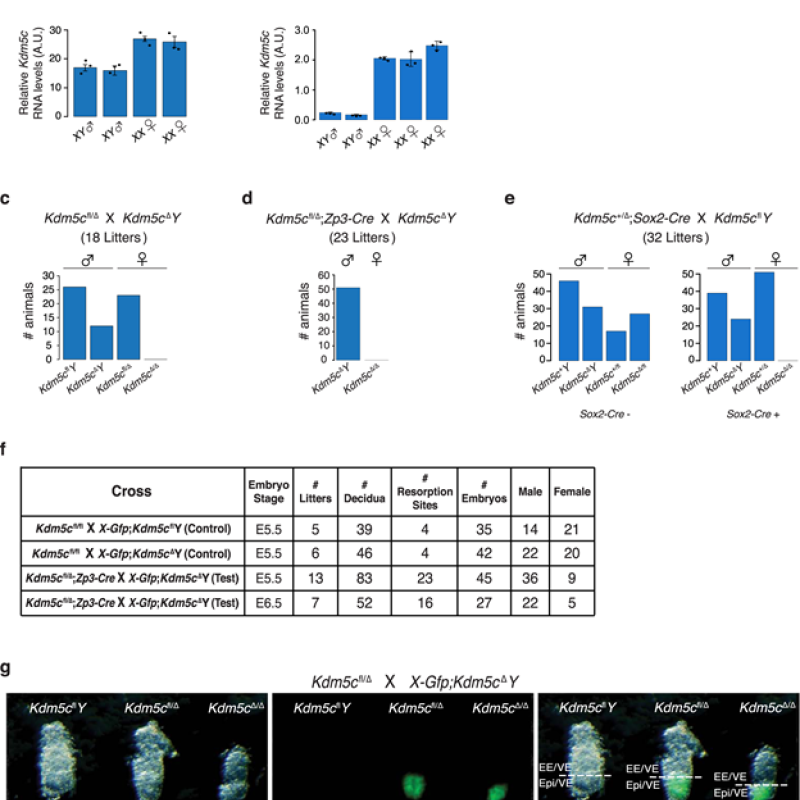
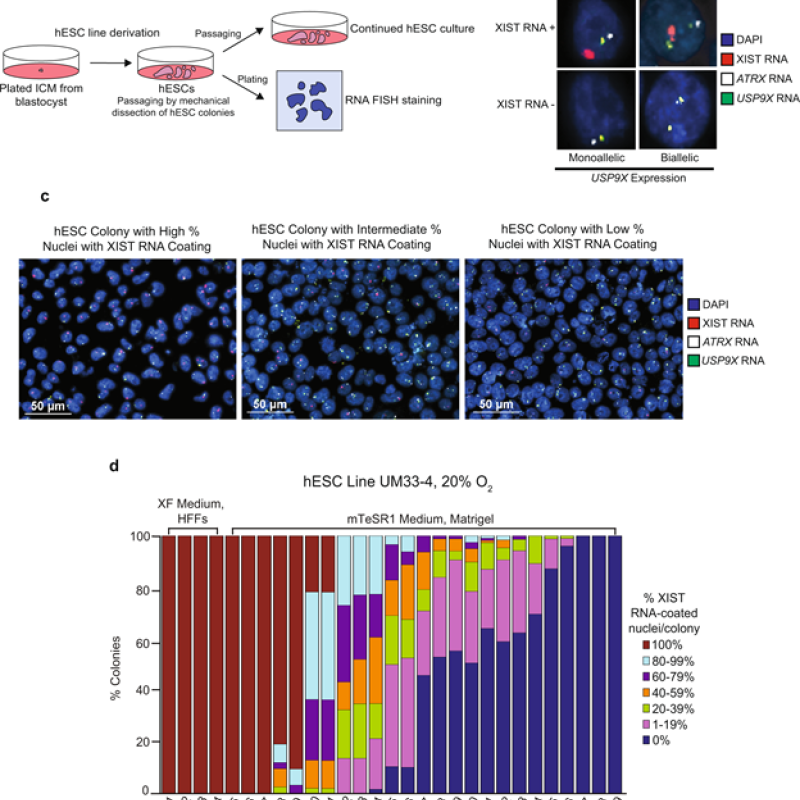
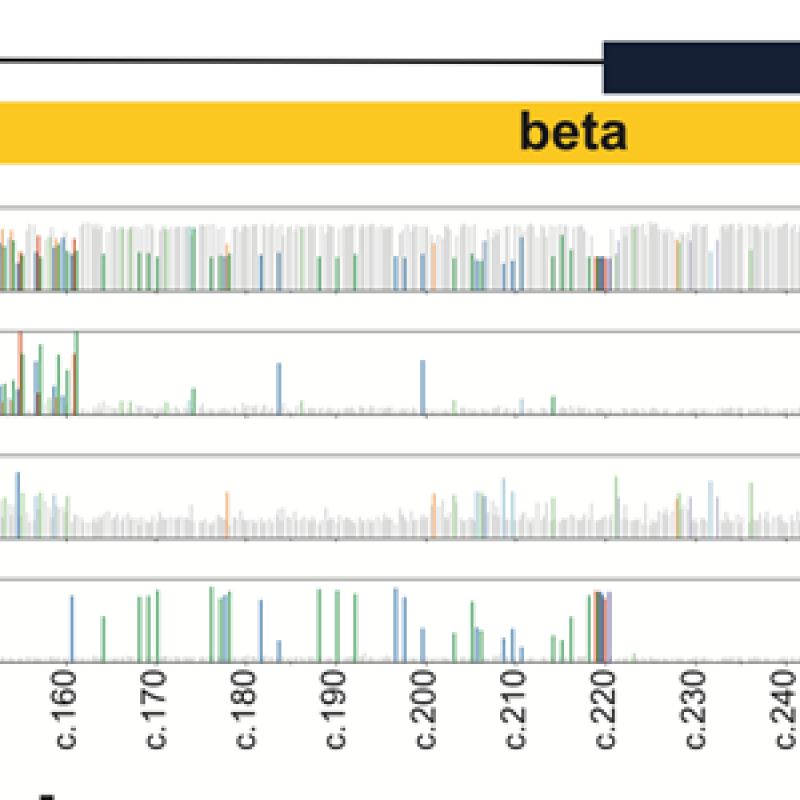
Stephen C.J. Parker, Ph.D., is the recipient of the 2024 Outstanding Scientific Achievement Award from the American Diabetes Association (ADA)
“The diligent work of these passionate professionals is moving us closer to a world free of diabetes," said Charles “Chuck” Henderson, CEO of the ADA