NEPTUNE
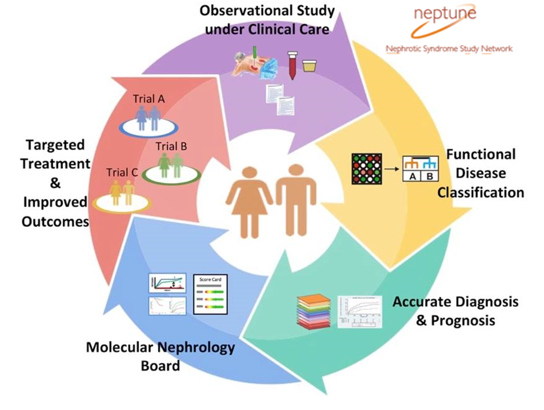
The Nephrotic Syndrome Study Network (NEPTUNE) is a 24-site consortium designed to foster clinical and translational research to redefine nephrotic syndrome on a mechanistic level and to translate new discoveries to the bedside. The NIH sponsored consortium is coordinated at the University of Michigan with a focus on patients with Focal Segmental Glomerulosclerosis (FSGS), Minimal Change Disease (MCD), Membranous Nephropathy (MN) or childhood onset nephrotic syndrome.
NEPTUNE includes a) longitudinal cohort study with biospecimens and data collection for use in scientific discovery and b) a biology-based patient to clinical trial matching program, NEPTUNE MATCH. Learn more at https://neptune-study.org.

CureGN
Glomerulonephropathy (CureGN) is a 66-center consortium that fosters research through a prospective observational study and biobank with 2,400 patients with glomerular disease.
Funded by the NIH-NIDDK, University of Michigan manages the CureGN data coordinating center. Participating children and adults with FSGS, MCD, MN, or IgA nephropathy graciously share their clinical data, patient-reported data, biospecimens and insight to support clinical and translational research. Learn more at https://curegn.org.
Kidney Research Network [KRN]
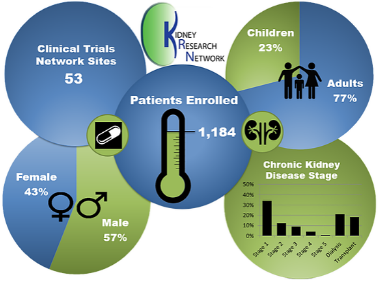
The mission of the KRN is to improve kidney disease treatment options and optimize patient health. KRN engages patients, medical teams, researchers, patient advocates, regulatory, and industry partners to accelerate research to find better treatments with a focus on rare kidney diseases.
Coordinated by the University of Michigan Pediatric Nephrology Research Program, the KRN works toward this mission in several ways. An electronic health record Registry of about 1,200 participants with glomerular kidney disease supports work across the other cores which include an Endpoints and Outcomes Consortium; Quality Improvement Core; Clinical Trials Consulting Team, and Clinical Trials Network. Learn more at https://www.kidneyresearchnetwork.org

FSGS Recurrence Collaborative
The FSGS recurrence collaborative is an effort to advance the understanding of recurrent focal segmental glomerulosclerosis (FSGS) and advance therapeutic options. The collaborative aims to foster and accelerate bench to bedside research focused on FSGS recurrence by
- Convening trans-disciplinary investigators to participate in an assessment of FSGS recurrence
- Establishing a prospective cohort study and biobank
- Developing a deeper understanding of disease epidemiology and mechanisms
- Identifying and prioritizing state of the art methods and projects to accelerate mechanism and management discovery
Learn more at https://www.fsgsrecurrencecollab.org/
Chronic Kidney Disease in Children (CKiD)

The CKiD Study is a NIH-funded, multicenter, prospective cohort study of children aged 6 months to 16 years with mild to moderate impaired kidney function.
The primary goals of CKiD are to determine the risk factors for decline in renal function and to define how progressive decline in renal function impacts biomarkers of risk factors for cardiovascular disease; neurocognitive function and behavior; and growth failure and its associated morbidity. Learn more at https://statepi.jhsph.edu/ckid.
Differential practice patterns in neonatal kidney failure
Renal replacement therapy in newborns with hyperammonemia due to inborn errors of metabolism
*Each of the noted studies above encourage ancillary studies, where investigators both within and outside the research consortia may request access to data or samples to address specific research questions.



